Question #158f3
1 Answer
Intermolecular bonds are caused by the attractive forces between the negative end of one molecule and the positive end of another.
DIPOLE-DIPOLE BONDS
A polar molecule has a positive end and a negative end. When two polar molecules are near each other, they arrange themselves so that the negative and positive ends line up and attract the two molecules together
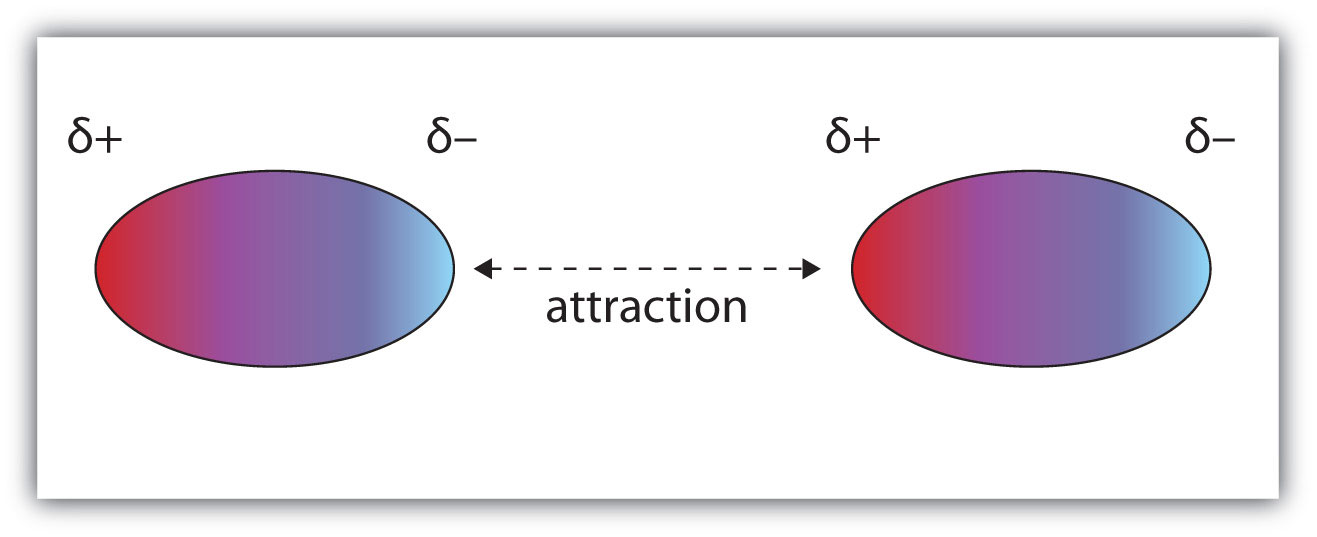
A hydrogen bond is the dipole-dipole attraction between the positive ends (the H atoms) of the O-H, N-H, and F-H bonds in one molecule and the negative ends (the N, O, or F atoms) in a neighbouring molecule.
In liquid water, for example, every water molecule can be H-bonded to four other water molecules.

There are other, weaker, attractive forces, but dipole-dipole attractions are the strongest attractions among covalent molecules.

