Question #7216f
1 Answer
A moving electron is an electric current and every electric current carries a magnetic field.
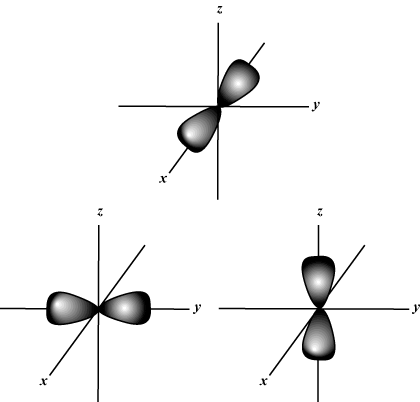
Consider the p orbitals which align themselves along the x, y and z axis. They are said to be * degenerate* ** which means they are of equal energy.
If a strong magnetic field is applied this causes the orbitals to have slightly different energies depending on whether they are aligned with the field or against it.
The principle quantum number is
The angular momentum quantum number is
The magnetic quantum number, to which your question refers is
The next quantum number is
As an example suppose
So
0 corresponds to 2s and 1 corresponds to 2p
An important principle was put forward by Pauli which states that in an atom, no 2 electrons can have all 4 quantum numbers the same.
This forms the basis by which atoms are built up.


